 By Avinash Shanbhag and Jeff Smith, ONC
By Avinash Shanbhag and Jeff Smith, ONC
Twitter: @ONC_HealthIT
As part of ONC’s ongoing charge to coordinate across federal and industry stakeholders, we determined it was necessary to adjust our Standards Version Advancement Process (SVAP) timeline. Although it may seem like this process has been around for a while, it’s still brand new and we’ve been looking at ways to optimize how the process aligns with other standards development work in the community. The changes we’ve made will help ensure timely publication of implementation specifications central to our cadence for new versions of USCDI.
Back in 2020, as part of the ONC Cures Act Final Rule, ONC established an annual Standards Version Advancement Process to enable health IT developers in the ONC Health IT Certification Program to voluntarily update their products to incorporate national coordinator-approved, newer versions of adopted standards and implementation specifications. This supports certified health IT developers and permits them to adopt and implement newer standards versions and support their clinical customers in nationwide, interoperable health information exchange, via this ongoing, collaborative process.
While the standards community has enthusiastically supported the SVAP concept, its real-world use is new. ONC’s Health IT Certification Program includes dozens of standards developed by numerous organizations with their own standards development activities and timelines. Additionally, ONC has finalized the first new version of the United States Core Data for Interoperability (USCDI) version 2 and initiated the process to identify USCDI version 3.
New SVAP Timeline
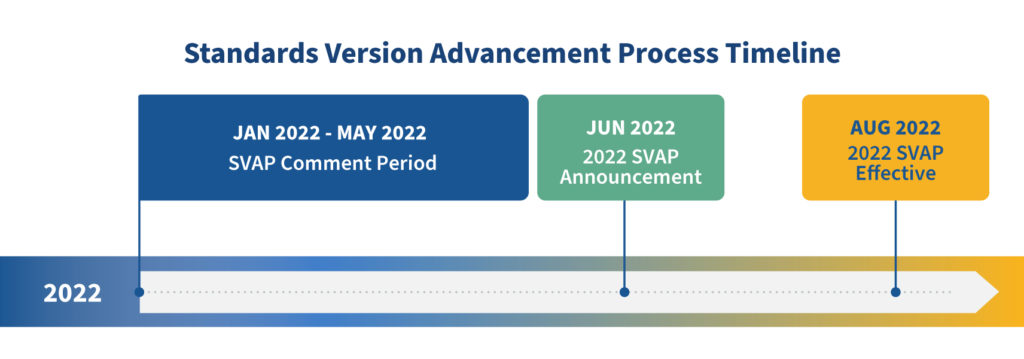
As indicated by HL7, newer versions of the US Core Implementation Guide supporting the latest version of USCDI are planned for publication in March of the following year. We expect this cadence of standards development activity will continue in future years, so beginning with the current SVAP and continuing in future years, ONC will announce approved standards for SVAP in June each year and anticipate them becoming effective for Certification Program use in August of that year. The June SVAP announcement and August effective date will be supported by a comment period from January to May each year.
Change to Current SVAP
The most immediate change for the public to note is that the current SVAP comment period, due to end September 30th, was extended to May 2, 2022. Additionally, the announcement of next version of approved standards for SVAP will be delayed by approximately 6 months, from January 2022 to June 2022. We don’t expect this to cause any delays in implementation by health IT developers, but rather the extra time will benefit in further maturing the implementation specifications through additional testing and pilot activities.
Minor Changes to Naming Convention for Future SVAP
We had previously named SVAP releases (e.g. Approved Standards for 2020) based on the year during which the comments were received even though the actual SVAP announcement was made the following year.
With the updated timeline, and since the entire SVAP process will conclude within the same calendar year, we have taken the opportunity to reset the naming convention to match the year in which the SVAP is approved by the national coordinator. Hence, the next release of standards approved via SVAP will be named as “Approved Standards for 2022” instead of 2021. (We anticipate a future “trivia quiz” question about missing SVAP release for 2021!)
This article was originally published on the Health IT Buzz and is syndicated here with permission.
